
ConclusionĮmulsions are mixtures of two or more liquids that are typically incompatible with one another. 
Small droplets of a polymer binder liquid are stretched out without dissolving in water to create these spheres. 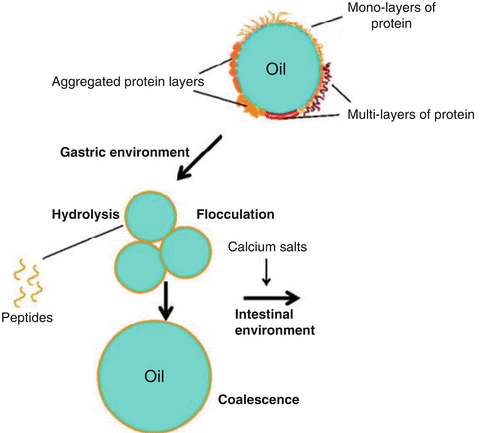
For example, pigments are encased in small polymer particles in emulsion paint. The vehicle refers to both the binder and the solvent as a unit. The colour, binder, and solvent are the three essential components of most paints. While the compositions of paints created for different purposes will change, they will share several key qualities. To untrained eyes, paints may appear to be nothing more than a colourant dispersed in a fluid to facilitate application. Lecithin is an example of a natural preservative (found in egg yolks). However, commercial salad dressings are often created with additives and emulsifying agents to prevent the vinaigrette from separating. Technically, homemade vinaigrettes are transient emulsions because the oil and water droplets are chemically separated and will soon re-form a different mixture. Because of this, espresso is nothing more than an emulsified liquid. The espresso machine provides the necessary pressure to combine the oils with the water because oils and water do not mix. In addition to providing flavour and aroma, coffee beans are also rich in oils. There’s indeed more to it than meets the eye, however. As a result of the machinery necessary to create it, many of us acquired this word. Oil droplets are suspended in a yolk, lime juice, or vinegar-based emulsion, giving both flavour and stabilisation.Ĭoffee’s global ubiquity has spawned a slew of creatively called subvarieties. This is a good illustration of an oil-in-water emulsion. They don’t form separate layers because they’re shielded by a membrane layer protecting the fat particles from colliding with water. 5 Real-life Emulsion Examplesīelow mentioned are five real-life examples of emulsion:Īn emulsion of fat globules (particles) distributed in water (aqueous) is milk. Milk is an excellent oil-in-water emulsion example. In an oil-in-water emulsion, the oil is the dispersed portion, and water is the dispersion medium or continuous segment. Examples include cod liver oil, margarine and so forth. Emulsions of this type are commonly found in butter and cold cream. Oil emulsions are another term for these emulsions. 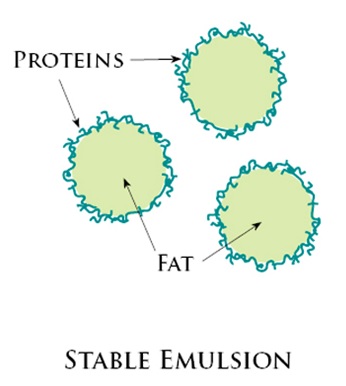
In these emulsions, the water is disseminated, and oil serves as the distribution medium. There are many additional examples of emulsions, including butter, which is the dispersion of water in fat and lecithin-rich egg yolk in the form of butter.Ĭlassifying emulsions is based on the characteristics of the dispersed segment and medium.The photographic emulsion, for example, is a gelatin gel with microscopic crystals distributed throughout it.A collection of mixed systems known as solutions, gels, or suspensions is also called an emulsion.As a result, drops of oil are formed and then dispersed in the water. When oil and water combinations are shaken together, they form emulsions.The dispersed phase and the dispersion medium are liquids in an emulsion, which can be explained in the simplest terms possible: The dispersion medium is one of the liquids, while the dispersed phase is the other. How are Emulsions Formed?Ī dispersion of two immiscible liquids is the basic building block of an emulsion. On the other hand, butter is when particles or droplets of a water-based solution get dispersed in fat. Milk is when fat molecules get dispersed in a water-based solution. In most cases, such mixtures are created by mixing liquids that are not soluble in one another, either in their natural form or with mechanisms like agitation.Įxamples of emulsions include milk and butter. In emulsions, two or more liquids are mixed in such a way that the droplets of one of the liquids are dispersed throughout the other.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
